HPLC Solvent Recycling
Reduction of HPLC Waste Solvent Using Spinning Band Distillation
HPLCs use solvents or mixtures of solvents as the mobile phase. Once used, the disposal of these solvents presents an ever-increasing problem. Not only is there the expense of disposal, but also the cost of managing and storing the waste. Spinning band distillation can be used in a number of ways to reduce the amount of waste solvent from HPLCs.
Get the Water Out!
Most HPLC waste contains large amounts of water, up to 90%. So for every liter of solvent purchased, there is up to 10 liters of waste generated!
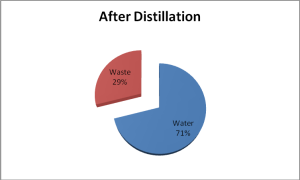
Removing the majority of water from these wastes is easily accomplished using spinning band distillation. Solvents used in HPLCs have lower boiling points than water. When distilled, these solvents boil /. The water is left behind in the boiling flask flask. This water is suitable for drain disposal.
Consider a common acetonitrile waste containing 75% water and 25% acetonitrile. If there are 10 liters of this waste there would be 7.5 liters of water and 2.5 liters of acetonitrile.
Spinning band distillation of this waste would yield approximately 2.9 liters of the acetonitrile/water azeotrope (87% ACN/13% water).
The 7.1 liters remaining in the boiling flask will be water free of acetonitrile that can be disposed of down the drain.
So instead of having 10 liters of waste hauled away, only 2.9 liters would need to be disposed. This represents a savings of 71%!
Water removal is the best and fastest way to reduce HPLC waste disposal. It requires very little effort to manage and operate the recycling equipment while producing the largest share of savings.
9600 Spinning Band Solvent Recycler
- Distillation Column Type: Spinning Band
- Throughput: 2-3 liters per hour
- Batch Sizes: 5, 12, 22 and 50 liter
- Contact Surfaces: Glass and Teflon
| Contact Us |
|

What about the acetonitrile I distilled?
The distilled acetonitrile is normally mixed with 13% water due to the azeotrope that forms between acetonitrile and water. Sadly, it is not possible to recover acetonitrile that contains less water.
